Is Gene Editing the End of GMO’s?

Sustainability Through Regenerative Organic Agriculture
June 30, 2020
COTA’s Organic Week Campaign Delivers New Creative and Strong Support From Industry in 2020
September 30, 2020Written by Guest Contributor Lucy Sharratt, Canadian Biotechnology Action Network (CBAN)
There is a lot of excitement in the media and research community about “gene editing” or genome editing. The techniques are being widely described as precise and, in some cases, as non-GMO. Neither is correct.
After over twenty years dominated by consumer controversy and largely limited to herbicide-tolerant traits (88% of all the worlds GM crop acres), the biotechnology industry has new techniques and a renewed promise of new products, and new public relations. However, just like with first-generation genetic engineering (genetic modification or GM), genome editing techniques are moving quickly in the lab to create new GMOs, even while our knowledge about how genomes work remains incomplete. The techniques are powerful and speedy but can be imprecise, leading to unexpected consequences.
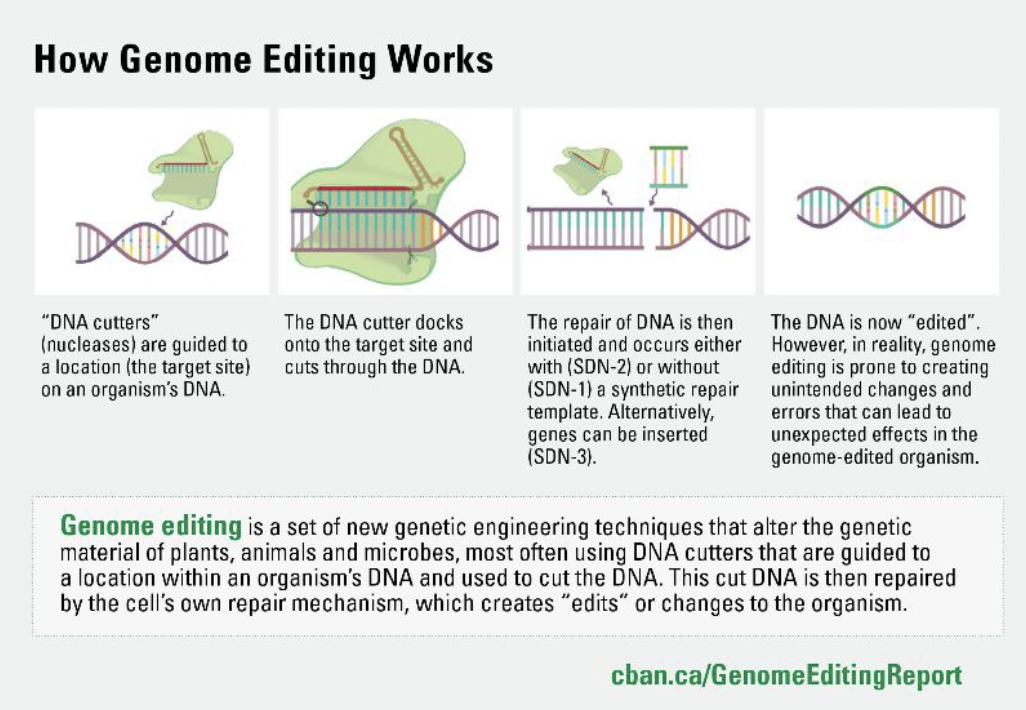
What is genome editing?
Genome editing techniques can be used to alter the genetic material of plants, animals and other organisms. They aim to insert, delete or otherwise change a DNA sequence at a specific, targeted site in the genome. Genome editing techniques are a type of genetic engineering, resulting in the creation of genetically modified organisms (GMOs).
The genome is the entire set of genetic material in an organism, including DNA.
Genome editing most often uses DNA cutters that are guided to a location within an organism’s DNA and used to cut the DNA. This cut DNA is then repaired by the cell’s own repair mechanism, which creates “edits” or changes to the organism. The most frequently used genome editing technique is called CRISPR, but other techniques follow similar principles.
First-generation genetic engineering techniques insert genes, at random locations, to permanently become part of the host organism’s genome, creating new DNA sequences. In contrast, new genome editing techniques insert genetic material that is then guided to a specific target site to perform “edits.” This means that, with genome editing, the inserted genetic material makes changes to the genome but does not necessarily have to become incorporated into the resulting GMO. This means that some genome-edited GMOs will not be transgenic.
Unexpected and unpredictable effects
Genome editing can be imprecise, and cause unexpected and unpredictable effects. Many studies have now shown that genome editing can create genetic errors (such as “off-target” and “on-target” effects). The much discussed CRISPR-Cas9 system, for example, can make unintended edits to the host’s DNA at additional sites to the target location. Such errors can lead to unexpected and unpredictable outcomes, such as changes in protein composition, in the resulting GMO.
Genome editing techniques can create unintended changes to genes that were not the target of the editing system (off-target effects), and even where a technique succeeds in making the intended change at the target location, it can also lead to other unexpected outcomes (on-target effects).
Genome editing can inadvertently cause extensive deletions and complex re-arrangements of DNA. Furthermore, during some genome editing processes, unwanted DNA can unexpectedly integrate into the host organism. To further complicate the question of risk, there are no standard protocols yet to detect off-target and on-target effects. Sometimes intended changes being created by genome editing techniques are described as “mutations” because only very small parts of DNA are altered and no novel genes have been intentionally introduced. However, even small changes in a DNA sequence can have big effects.
The functioning of genes is coordinated by a complex regulatory network that is still poorly understood. This means that it is not possible to predict the nature and consequences of all the interactions between altered genetic material and other genes within an organism. For example, one small genetic change can impact an organism’s ability to express or suppress other genes.
And End to GMO Regulation?
Despite these risks, one of the more attractive aspects of using genome editing appears to be the prospect of less regulation. Products of genome editing will be regulated in Canada but, according to a recent survey of Canadian plant breeders, one of the main advantages of using genome editing in research is “the potential for easier regulatory paths to commercialization.”(1) This easier path is largely an assumption but without increased transparency in Canadian regulation, exactly how products will be assessed is unknown.
The new techniques of genome editing will challenge regulators with new traits and new processes, with increasing complexity and ongoing uncertainty. This means that, rather than assume safety, the new technologies need to be met with precaution and increased independent scrutiny.
The most fundamental question – do we need to use this technology? – remains unasked in regulation, but it is answered by organic farmers; an answer that is embraced by many Canadians.
New Report
For more information and discussion about genome editing, read the new report from the Canadian Biotechnology Action Network “Genome Editing in Food and Farming: Risks and Unexpected Consequences.”
The report and an introductory factsheet are available online at: www.cban.ca/GenomeEditingReportFor the report in French, stay tuned to www.rcab.ca/edition-genome
For updates or to find out more, visit: www.cban.ca/genome-editing.
(1) Stuart Smyth, Is gene editing being properly used? The Western Producer, April 9, 2020.
 About Lucy Sharratt:
About Lucy Sharratt:
Lucy Sharratt is the Coordinator of the Canadian Biotechnology Action Network (CBAN) which brings together 16 groups to research, monitor and raise awareness about issues relating to genetic engineering in food and farming. CBAN members include farmer associations, environmental and social justice organizations, and regional coalitions of grassroots groups. CBAN is a project on Make Way’s shared platform.
Learn more about CBAN here.






